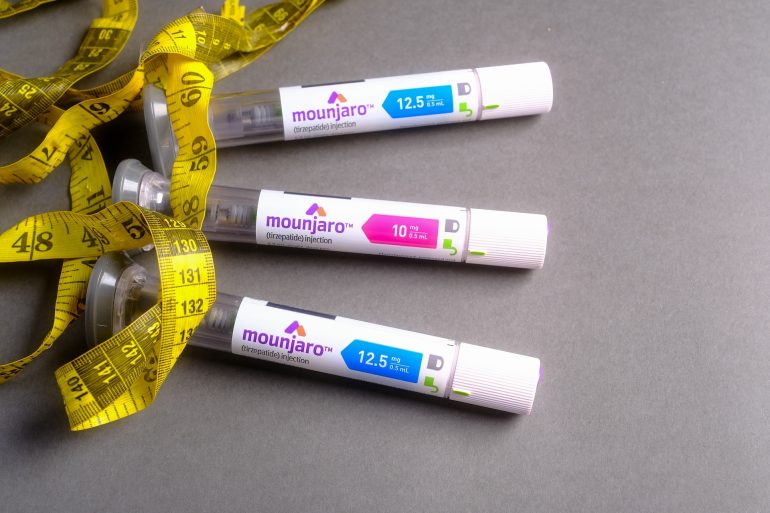
DKSH Singapore has secured approval of Mounjaro® (tirzepatide) KwikPen® on 18 June 2025, a once-weekly, prescription-only medicine shown to improve blood sugar control and support clinically meaningful weight loss.
Type 2 diabetes and obesity continue to rise here, and many patients still struggle to reach targets despite best efforts with diet, exercise and existing therapies. This approval answers a clear unmet need and aligns squarely with Singapore’s broader health push to tackle metabolic illness at scale.
In practical terms, Mounjaro® (tirzepatide) KwikPen® is indicated for adults with insufficiently controlled type 2 diabetes mellitus, either as monotherapy when metformin is unsuitable, or in combination with other anti-diabetic medicines.
It is also approved as an adjunct to a reduced-calorie diet and increased physical activity for weight management, including weight loss and weight maintenance, in adults with an initial BMI ≥30 kg/m² (obesity), or BMI ≥27 to <30 kg/m² (overweight) in the presence of at least one weight-related comorbidity such as:
- Hypertension
- Dyslipidaemia
- Obstructive sleep apnoea
- Cardiovascular disease
- Prediabetes
- Type 2 diabetes mellitus.
Tirzepatide is a dual GIP and GLP-1 receptor agonist. In essence, it activates two natural hormone pathways that influence insulin secretion, appetite regulation and post-meal glucose control. The combination aims to deliver a one-two punch: steadier sugars and reduced caloric intake.
Across pivotal clinical trials, tirzepatide demonstrated clinically meaningful weight loss alongside improvements in glycaemic control, a result that has drawn interest from both endocrinologists and primary care doctors looking to simplify complex regimens.
The delivery is intentionally straightforward: a once-weekly KwikPen® designed to fit real life, not complicate it. For many patients juggling shift work, childcare and the rest of life’s noise, fewer injections can mean better adherence, which, in turn, can translate into better outcomes.
Of course, medicines are only part of the story; lifestyle remains the foundation and beams of the house. But when you reinforce those beams with modern pharmacology, the structure tends to hold.
On the partnership front, Eli Lilly and Company has appointed DKSH Singapore as marketing authorisation holder, granting DKSH distribution and promotion rights for Mounjaro® (tirzepatide) KwikPen® locally.
Will it solve metabolic disease on its own? Of course not. But for thousands of Singaporeans sitting on the cusp, this is timely, targeted help. The next lap belongs to clinics, counsellors and patients, working together with a clearer line to the chequered flag.
Check out more news articles on Futr here!